Strides Pharma to conduct human studies of Favipiravir: R Ananth, CEO & MD
R Ananthanarayanan, CEO & MD, Strides Pharma, talks about what led to one-time loss and margin contraction in March quarter, launches in FY21.
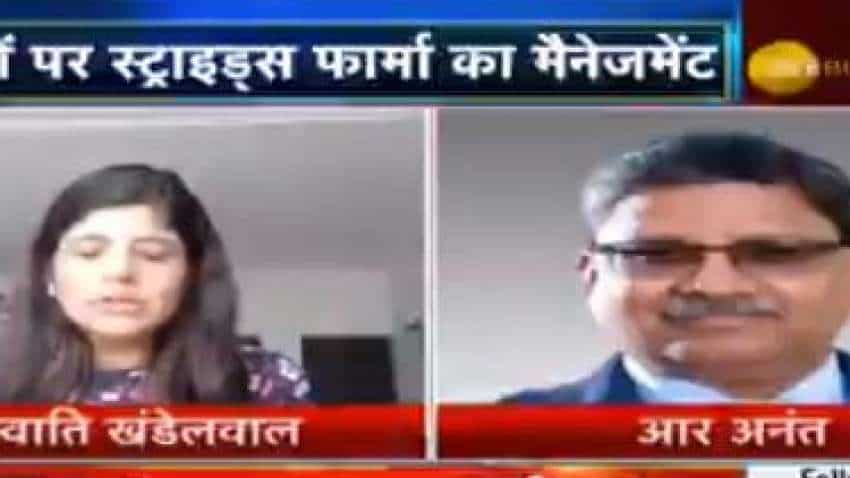
R Ananthanarayanan, CEO & MD, Strides Pharma, talks about what led to one-time loss and margin contraction in March quarter, launches in FY21 and human study of Favipiravir during an exclusive interview with Swati Khandelwal, Zee Business. Edited Excerpts:
Strides pharma has seen a one-time loss of Rs 170 crore loss in the March quarter and there is a decline in margin and income. What led to this one-time loss and as well as margin contraction?
Strides Pharma faced a setback in the fourth quarter and after the USFDA requested all manufacturers including us to withdraw Ranitidine products from the US market. The fourth quarter got impacted clearly due to Ranitidine withdrawal from the market. This was a one-time exception. If you have a look at the complete year than it was fantastic and we have met significant milestones. The fourth quarter was impacted due to Ranitidine withdrawal due to which we have to take a write-down of Rs 170 crore of which Rs 124 crore relates to Ranitidine impact. The other Rs 52-55 crore comes from the mark-to-market impact on our Australia deferred the receivables, which is a notional value because the conversion of the currency is showing it has returned. Therefore the real impact is of Rs 124 crore coming from Ranitidine.
See Zee Business Live TV streaming below:
Let us know about the planned launches for FY21, especially the major launch that we should look out for?
This is a great point and I would like to spend little time explaining that because our US business is flatted to grow significantly even without benefitting. If you take things without taking Ranitidine numbers in the US for the entire year, it comes to about $192 million. Now, our expectation and confidence are that we will grow by 25-30% over the $192 million, which is without Ranitidine. For getting to this 25-30% growth our levers are as follows: (i) New product launches. The new product launches that we did in FY20, we had launched about 6 products, which delivered around $20 million and the full-year impact of those launches will come around $45 million, which is factored for FY21. (ii) We also have a set of 25 products which has already been approved by USFDA but are not commercialized. We have already started working towards getting the spiked chain, getting the product ready, scale-up and manufacturing. We will be able to launch the products and have been choosy in selecting the products that meet our financial threshold on our margins. (iii) In the US, we have a BA program, which supplies to government contracts. Last year, we did two products under the program. This year, we are set to launch 5 plus products for the program. And, two of our facilities have qualified as designated plants for supply. Combination of all of these will allow us to grow our US business by 25-30% over the $192 million, which is without Ranitidine.
Tell us about your strategy for the emerging market and are you looking towards new geographies that you want to entre?
When it comes to emerging markets than in the last few quarters we have seen a decline in our revenues. It was a result of the design as we are doing a reset in our strategy, which was to move away from products that were low margin and have a healthy mix of products that is better in the margin. Now, we are done on reset and are ready to execute on select orders that meet our margin threshold. The second element in the emerging market is institutional business. On the institutional business, of course, this is a COVID scenario, it is always a tendency of the global tenders to start focusing on COVID rather than focus on other areas of ARB, Malaria and so on. So, we will need to do a little bit wait and watch. We have some products in our portfolio but there is a need to do a bit of watch. Overall on emerging market and institutional, we do this to be almost bottomed out in FY20 and we expect it to start showing improvement through the year.
Can you update us on the phase in which the drug trial of Favipiravir that was being conducted in India and commercialization plan for the same?
Favipiravir was announced in April and we have already commercialized the very complex product and manufactured it and started exporting the product out of India to the Gulf Cooperation Council (GCC) countries. We are also working expeditiously with the Indian authorities for us to get clearance for human studies. We got the approval for human studies but I want to clarify that when we say human study it means that we will be doing a bio-equivalent study in comparison directly with Avigan, the innovator product from Japan and will be completing that study and submitting the dossier for approval from the regulatory authorities.
09:36 AM IST





 COVID-19 Update: India reports 159 new coronavirus cases, 3 fresh deaths
COVID-19 Update: India reports 159 new coronavirus cases, 3 fresh deaths COVID-19 Update: India reports 441 new coronavirus cases, no fresh deaths
COVID-19 Update: India reports 441 new coronavirus cases, no fresh deaths Coronavirus news: 3,422 active COVID-19 cases recorded in India
Coronavirus news: 3,422 active COVID-19 cases recorded in India  COVID-19 Update: India records 774 new coronavirus cases
COVID-19 Update: India records 774 new coronavirus cases Coronavirus Update: India reported 109 cases of COVID subvariant JN.1 till Tuesday
Coronavirus Update: India reported 109 cases of COVID subvariant JN.1 till Tuesday