Lupin gets USFDA approval for generic Alzheimer treatment capsule
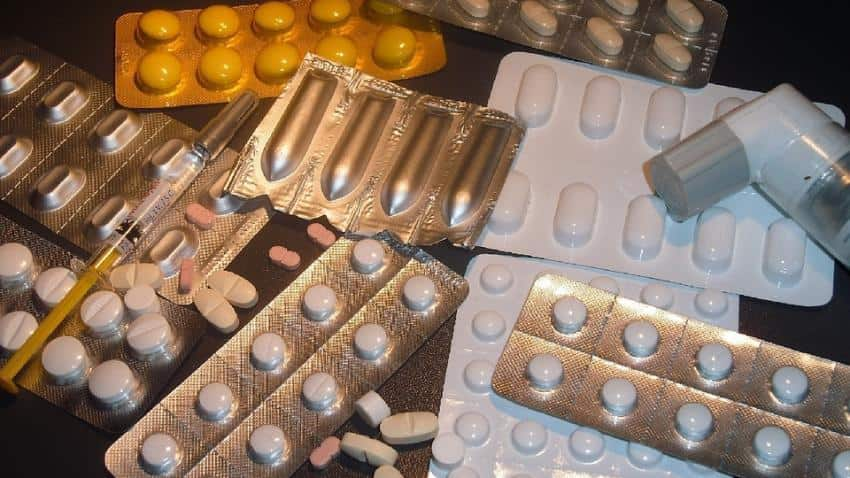
Pharma major Lupin on Thursday said it has received final approval from the US health regulator to market its memantine hydrochloride extended-release capsules, which are used in the treatment of the Alzheimer's disease.
"....the company has received final approval for its Memantine Hydrochloride Extended-Release Capsules, 7 mg, 14 mg, 21 mg, and 28 mg from the United States Food and Drug Administration (USFDA) to market a generic version of Allergan’s Namenda XR Capsules, 7 mg, 14 mg, and 28 mg," Lupin said in a filing to the BSE.
The company's product are the AB rated generic equivalent of Allergan’s Namenda Capsules, which are indicated for the treatment of moderate to severe dementia of the Alzheimer's type, it said.
According to IMS MAT June 2016 data, the Nameda XR Capsules had US sales of $1.22 billion (nearly Rs 8,116.4 crore).
The stock of Lupin has closed down 3.10% or Rs 47.05 at Rs 1,469.50 on the BSE on Thursday.
Get Latest Business News, Stock Market Updates and Videos; Check your tax outgo through Income Tax Calculator and save money through our Personal Finance coverage. Check Business Breaking News Live on Zee Business Twitter and Facebook. Subscribe on YouTube.
RECOMMENDED STORIES
06:25 PM IST










 Lupin shares fall 2% despite receiving approval for Acotiamide tablets
Lupin shares fall 2% despite receiving approval for Acotiamide tablets Lupin to acquire 21 branded products from Japan's Shionogi
Lupin to acquire 21 branded products from Japan's Shionogi  Lupin's Q4 net profit up nearly 48%, stock up 1.42%
Lupin's Q4 net profit up nearly 48%, stock up 1.42%