Covid-19: Bharat Biotech's Covaxin, Serum Institute's Covishield vaccines get 'Conditional Market Authorization' approval form DCGI - details here!
DCGI on Thursday gave its nod for market authorization of two COVID-19 vaccines - Covaxin and Covishield subject to certain conditions. Consent came after the the Subject Expert Committee of CDSCO recommended for upgradation of status for vaccines from restricted use in emergency situations to grant of new drug permission
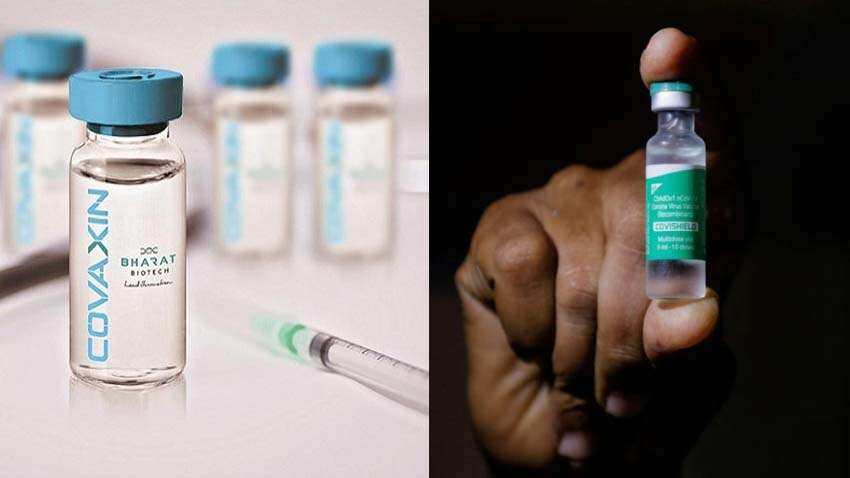
'Conditional Market Authorization' is a new category of market authorization that has emerged during the current global pandemic of COVID19





 India’s first intranasal Covid-19 vaccine trial over, Bharat Biotech calls it safe and immunogenic
India’s first intranasal Covid-19 vaccine trial over, Bharat Biotech calls it safe and immunogenic Covid-19 vaccine: Adults vaccinated with Covaxin, Covishield can take Corbevax precaution dose
Covid-19 vaccine: Adults vaccinated with Covaxin, Covishield can take Corbevax precaution dose  Vaccination for children: DGCI approves Covaxin for restricted emergency use in children aged 6-12 years
Vaccination for children: DGCI approves Covaxin for restricted emergency use in children aged 6-12 years Covid19: After getting regular market approval, each dose of Covishield, Covaxin likely to cost this
Covid19: After getting regular market approval, each dose of Covishield, Covaxin likely to cost this Government expert panel recommends regular market approval for Covishield, Covaxin
Government expert panel recommends regular market approval for Covishield, Covaxin