Coronavirus Vaccine Covishield: Side effects, Benefits of Serum Institute-AstraZeneca vaccination
Coronavirus Vaccine News: After India's drugs regulator, Drugs Controller General of India (DCGI) on Sunday approved Oxford COVID-19 vaccine Covishield, manufactured by the Serum Institute, a lot of questions are doing the rounds about various aspects of the coronavirus vaccine.
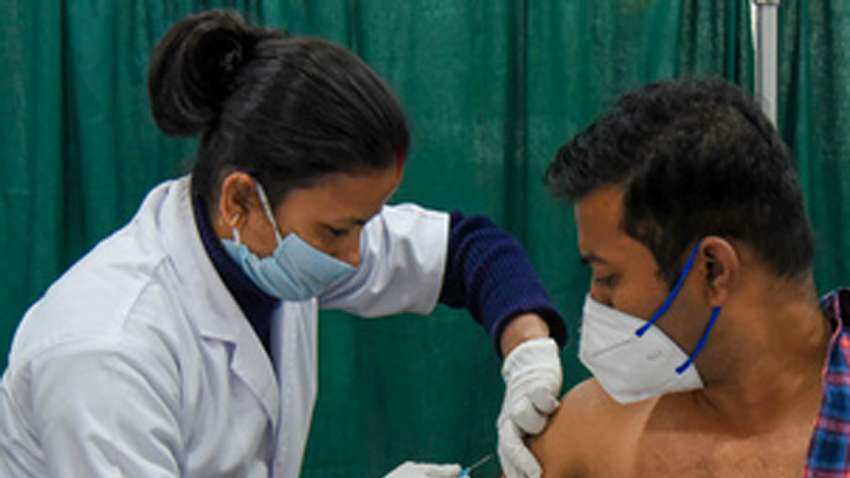
Coronavirus Vaccine News: After India's drugs regulator, Drugs Controller General of India (DCGI) on Sunday approved Oxford COVID-19 vaccine Covishield, manufactured by the Serum Institute, a lot of questions are doing the rounds about various aspects of the coronavirus vaccine. The Serum Institute of India, the world's largest vaccine manufacturer, has tied up with AstraZeneca to manufacture Covishield.
Here are the answers to some of the most commonly asked questions of COVID-19 vaccine Covishield:-
-What is COVID-19 Vaccine AstraZeneca and what is it used for?
COVID-19 Vaccine AstraZeneca is a vaccine indicated for active immunisation of individuals 18 years of age and older for the prevention of coronavirus disease 2019 (COVID-19).
-How does COVID-19 Vaccine AstraZeneca work?
COVID-19 Vaccine AstraZeneca stimulates the body’s natural defences (immune system) and causes the body to produce its own protection (antibodies) against the virus. None of the ingredients in this vaccine can cause COVID-19.
-How is COVID-19 Vaccine AstraZeneca used?
The pharmaceutical form of this medicine is a solution for injection and the route of administration is intramuscular injection. COVID-19 Vaccine AstraZeneca will be given to you by an authorised practitioner as an intramuscular injection into the muscle at the top of the upper arm (deltoid muscle). You will receive 2 injections of COVID-19 Vaccine AstraZeneca, each of 0.5ml. You will be told when you need to return for your second injection of COVID-19 Vaccine AstraZeneca. The second injection can be given between 4 and 12 weeks after the first injection.
-What benefits of COVID-19 Vaccine AstraZeneca have been shown in studies?
In a pre-specified preliminary analysis, those who received the vaccine had a reduction in the rate of COVID-19 illness compared to those who received the control (30 cases of COVID-19 illness in the vaccinated group compared to 101 cases in the control group). These results were observed two weeks or more after the second dose in study participants with no evidence of prior SARS-CoV-2 infection.
A similar benefit was observed in participants who had one or more other medical conditions that increase the risk of severe COVID-19 disease, such as obesity, cardiovascular disorder, respiratory disease or diabetes.
-What are the possible side effects of COVID-19 Vaccine AstraZeneca?
The most common side effects with COVID-19 Vaccine AstraZeneca (which may affect more than 1 in 10 people) were tenderness, pain, warmth, redness, itching, swelling or bruising where the injection is given, generally feeling unwell, feeling tired (fatigue), chills or feeling feverish, headache, feeling sick (nausea), joint pain or muscle ache. In clinical studies, most side effects were mild to moderate in nature and resolved within a few days with some still present a week after vaccination.
02:24 PM IST





 Coronavirus Vaccine: Precaution dose to be available from April 10 to 18+ population; facility to be available at private centres
Coronavirus Vaccine: Precaution dose to be available from April 10 to 18+ population; facility to be available at private centres Zydus Cadila vaccine for Covid 19: World's first DNA-based vaccine! What we know so far about 3-DOSE vaccine that can be administered to children aged 12 years and above too
Zydus Cadila vaccine for Covid 19: World's first DNA-based vaccine! What we know so far about 3-DOSE vaccine that can be administered to children aged 12 years and above too Covid 19 vaccination for 18 to 44 years: Delhi, Maharashtra, Karnataka among states that suspended inoculation drive—check full list here
Covid 19 vaccination for 18 to 44 years: Delhi, Maharashtra, Karnataka among states that suspended inoculation drive—check full list here  REVEALED: 'Laga bhi diya...' - What PM Narendra Modi told nurse P Niveda after COVID-19 vaccine shot
REVEALED: 'Laga bhi diya...' - What PM Narendra Modi told nurse P Niveda after COVID-19 vaccine shot BEWARE! Fake Coronavirus vaccine multi-million dollar scam busted! China at it again?
BEWARE! Fake Coronavirus vaccine multi-million dollar scam busted! China at it again?