Dr Reddy's seeks DCGI nod for conducting phase-3 clinical trial of Russian COVID-19 vaccine Sputnik V
The Hyderabad-based Dr Reddy's Laboratories has applied to the Drugs Controller General of India for permission to conduct phase-3 human clinical trials of the Russian vaccine Sputnik V against COVID-19 in India, sources said.
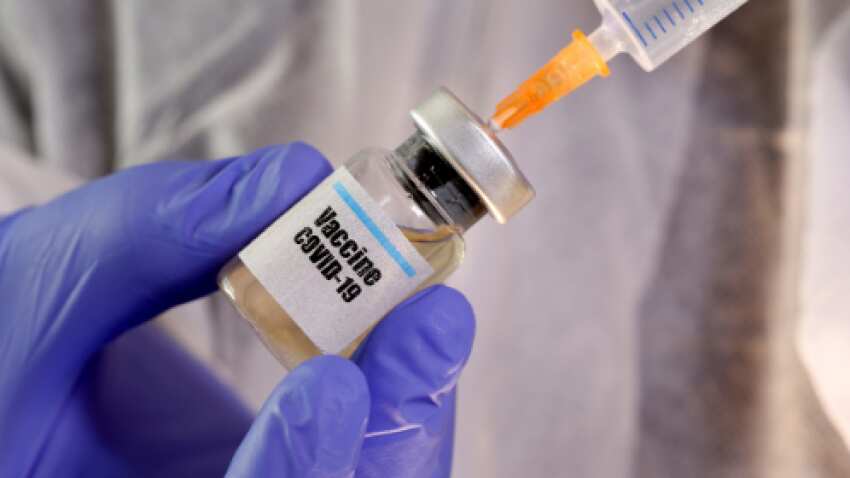
The Indian pharma giant has collaborated with the Russian Direct Investment Fund (RDIF) to conduct clinical trials of Sputnik V as well as its distribution. Photo: Reuters





 Dr Reddy's Labs enters into distribution partnership with Sanofi India; stock rises
Dr Reddy's Labs enters into distribution partnership with Sanofi India; stock rises  Dr Reddy's Labs hits an all-time high after US FDA issues EIR to its Hyderabad facility
Dr Reddy's Labs hits an all-time high after US FDA issues EIR to its Hyderabad facility Dr Reddy's Laboratories zooms to an all-time time after reporting above-estimate December quarter results
Dr Reddy's Laboratories zooms to an all-time time after reporting above-estimate December quarter results Dr Reddy's Labs Hyderabad facility gets 9 observations from US drug regulator; shares fall
Dr Reddy's Labs Hyderabad facility gets 9 observations from US drug regulator; shares fall Dr Reddy's Q4 2023 preview: Here is what to expect from the drug maker
Dr Reddy's Q4 2023 preview: Here is what to expect from the drug maker